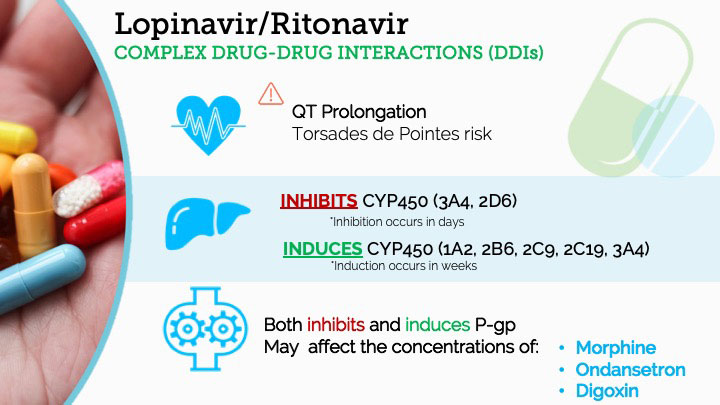
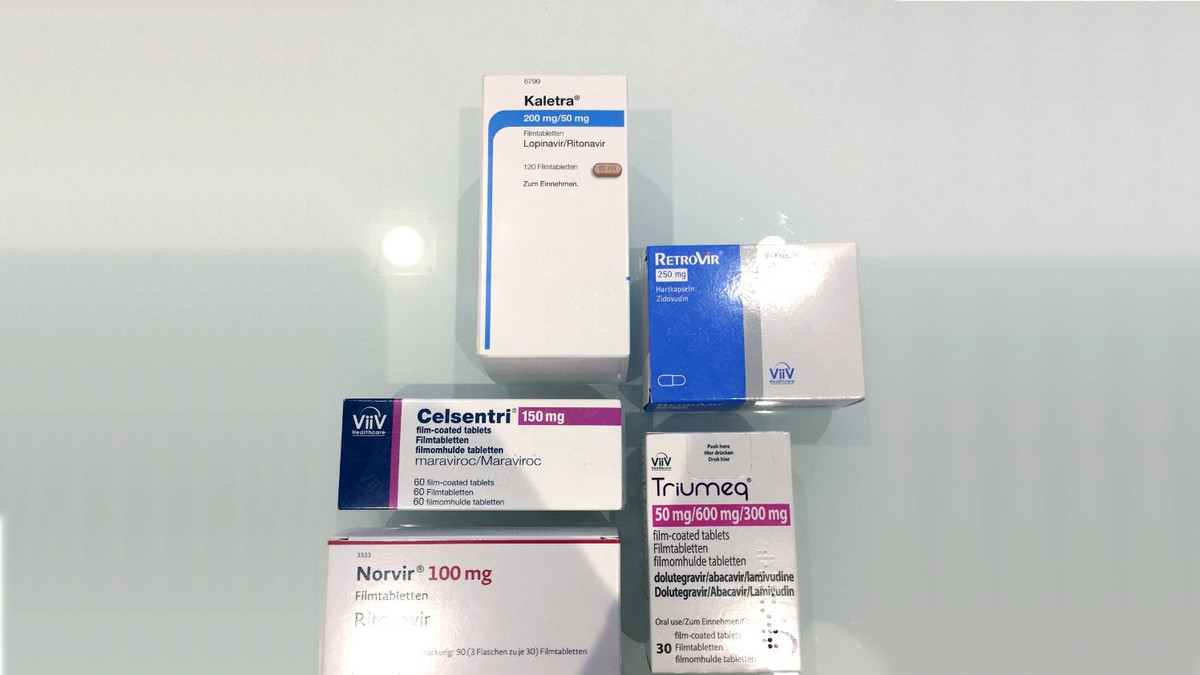
An overview of the safety assessment of medicines currently used in the COVID-19 disease treatment - ScienceDirect
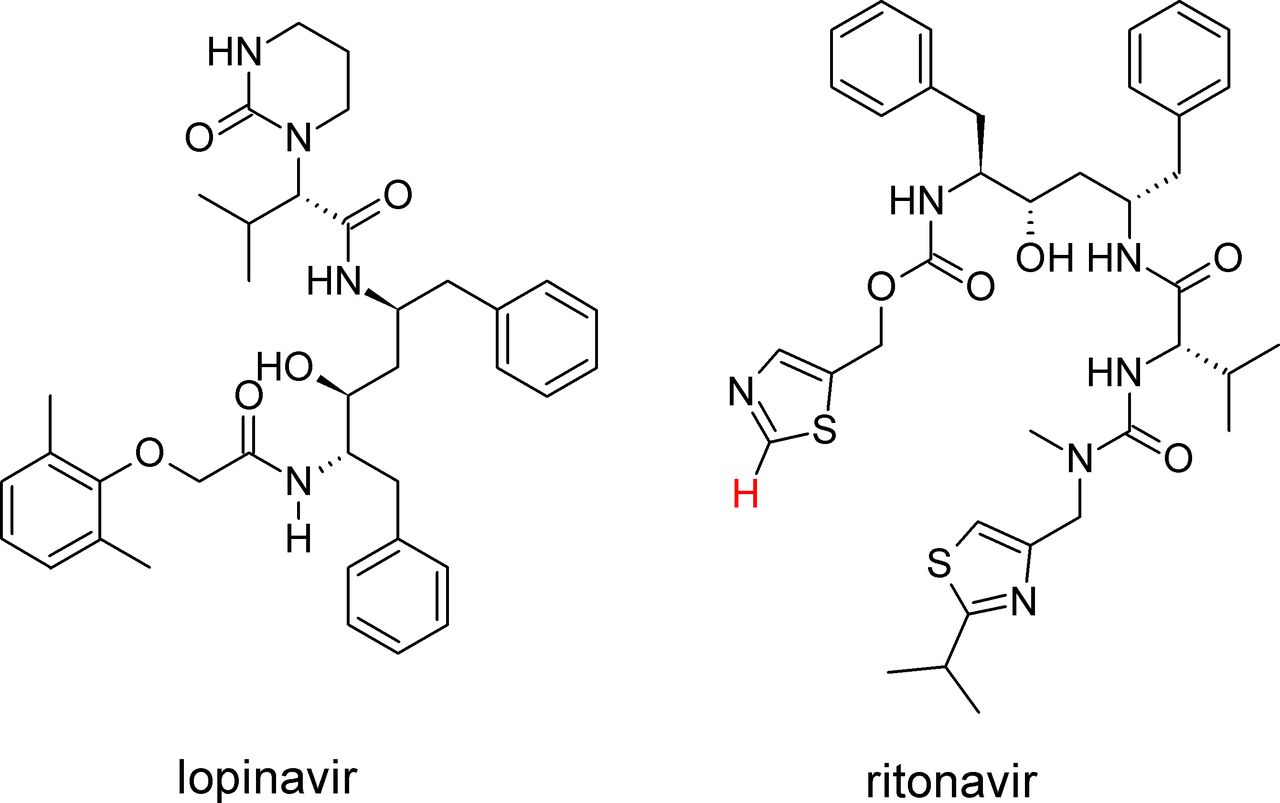
Lopinavir/ritonavir, a new galenic oral formulation from commercial solid form, fine-tuned by nuclear magnetic resonance spectroscopy | European Journal of Hospital Pharmacy
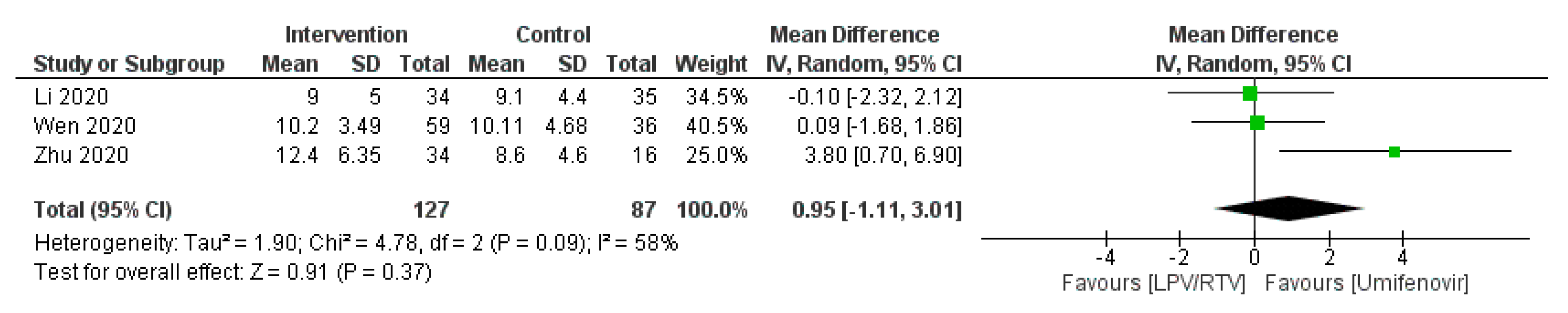
TropicalMed | Free Full-Text | Efficacy and Safety of Lopinavir/Ritonavir for Treatment of COVID-19: A Systematic Review and Meta-Analysis

Implications of the Digestion of Milk-Based Formulations for the Solubilization of Lopinavir/Ritonavir in a Combination Therapy | Molecular Pharmaceutics

Extended pre-exposure prophylaxis with lopinavir–ritonavir versus lamivudine to prevent HIV-1 transmission through breastfeeding up to 50 weeks in infants in Africa (ANRS 12174): a randomised controlled trial - The Lancet

Once-daily atazanavir/ritonavir versus twice-daily lopinavir/ritonavir, each in combination with tenofovir and emtricitabine, for management of antiretroviral-naive HIV-1-infected patients: 48 week efficacy and safety results of the CASTLE study - The ...

Lopinavir/ritonavir: A rapid review of effectiveness in COVID-19 - The Centre for Evidence-Based Medicine